| The Home page of ILPI's Safety Data Sheet (SDS) Resource, the leader in SDS information since 1995! | |
| The history and philosophy behind this resource. | |
| A curated collection of books and reference materials concerning Safety Data Sheets and closely related topics. | |
| Paste your plain text SDS into the SDS-Demystifier, and it will be converted into a hypertext-enriched document with links to detailed explanations of each key term. | |
| An extensive list of frequently asked questions about Safety Data Sheets including regulations, content, compliance, and more. | |
| A humorous take on Safety Data Sheet jargon. Fill in the blanks on our entry form to generate a personalized Unsafety Data Sheet to share with your coworkers. | |
| Since 1995, we've maintained this massive curated list of the best places to find Safety Data Sheets on the Internet. | |
| You are here! Way more than a glossary, this hypertext-enhanced resource covers hundreds of SDS-related terms and expert knowledge. Each entry includes both the SDS relevance and links to additional authoritative resources. | |
| Archived results of Safety Data Sheet related polls taken by some of our millions of site visitors | |
| The OSHA regulations behind SDS regulations, including the inspection guidelines and over 400 official interpretations letters under the Hazard Communication Standard | |
| Commercial suppliers of SDS authoring and management software as well as cloud compliance services. | |
| Commercial companies that will create SDS's for your specific needs as well as SDS translation companies. |

Safety signs, banners, and scoreboards? Get yours at Safety Emporium!

Store and dispense flammable liquids safely with flammable liquid safety cans from Safety Emporium.
Definition
An exothermic process is one that gives off heat. This heat is transferred to the surroundings.
An endothermic process is one in which heat has to be supplied to the system from the surroundings.
A thermoneutral process is one that neither requires heat from the surroundings nor gives off energy to the surroundings.
These terms are usually applied to chemical reactions. A chemical reaction can only be one of these three terms at any given moment. A reaction that is exothermic will be endothermic if run backward and vice-versa.
Additional Info
Exothermic chemical reactions liberate heat. A simple and familiar example is the combustion of methane gas (CH4). The balanced chemical reaction for this process is:
CH4(g) + 2 O2 (g) 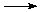 CO2(g) + 2 H2O(g).
CO2(g) + 2 H2O(g).
This process gives off a lot of energy, so we could write "heat" as one of the products on the right (products) side of the reaction if we wished. The term enthalpy, H, is used by chemists to describe how heat flows into or out of a system.
For an exothermic reaction, the change in enthalpy, ΔH, as we go from reactants (methane and oxygen) to products (carbon dioxide and water) is a negative quantity. For an endothermic reaction, ΔH is greater than zero. And for a thermoneutral reaction ΔH = 0.
ΔH tells us how much heat energy will be given off or will be required for a particular chemical reaction. It doesn't tell us whether the reaction will occur or how fast. A great example of strongly exothermic reaction that does not occur without a significant heat input to get it started is the thermite reaction which is so energetic that it can weld metal under water!
Endothermic reactions are usually not a great safety hazard. However, because the reaction draws heat from its surroundings, the reaction container may become cold and cause condensation or ice to form. This can be a safety hazard if the materials involved react with water.

Get your Class D and other specialty extinguishers from Safety Emporium.
SDS Relevance
These terms are not commonly found on Safety Data Sheets. If mentioned, exothermic is usually used in describing a sudden release of a large amount of heat in either Section 5 (fire-fighting measures), Section 6 (accidental release measures), or possibly Section 10 (stability and reactivity). Endothermic may be found similarly, but endothermic reactions are far less common.
Many common chemicals undergo exothermic reactions. For example, simply dissolving sodium hydroxide (NaOH) in water produces enough heat that if this is not done carefully it could melt a plastic container! Most mixtures of incompatible chemicals involve violently rapid exothermic reactions.
The heat that a chemical reaction gives off can quickly heat the surrounding area (or rest of the chemicals in the container) to very high temperature. As temperature increases, the rate of chemical reactions generally increase as well. Thus, once an exothermic reaction begins, it can quickly "run away" - accelerating its rate because of the heat produced. This can be especially dangerous if the material reaches its flash point or autoignition temperature, at which point a fire or explosion could occur. Therefore, always conduct a thorough risk assessment before performing (and especially before scaling up) an exothermic reaction.
Again, it is critically important to know when a chemical reaction can generate excess heat and to take appropriate measures to deal with this. Examples include slow mixing, using a cooling bath, or avoiding that reaction. When performing scaleups, be sure you carefully monitor the temperature, do not scale up too much in one leap, and prepare ahead of time for possible emergencies.
Further Reading
- A great example of what happens during an uncontrolled exothermic reaction.
- Examples of exothermic and endothermic processes at General Chemistry Online.
- Exothermic and Endothermic Reactions at Chemistry LibreTexts, an open-access textbook.
- Endothermic and Exothermic Reactions at ThoughtCo.
- WolframAlpha is a computational search engine that can return all kinds of chemical data, including thermodynamic properties.
See also: autoignition temperature, flash point, oxidation.