
Imido ligands, also referred to as imides or nitrenes, are a common ligands in high oxidation state transition metal chemistry. A metal imido has the general form M=NR where there is a double bond between the metal center and the nitrogen atom. In addition, the lone pair on nitrogen usually donates to the metal center as well, providing a three-bond interaction with the metal.
Chemists who work with high valent complexes usually consider the imido unit to be NR2- whereas those that work with low valent complexes tend to consider it a neutral ligand. When specifying how many electrons an imido ligand donates it is therefore necessary to state which electron counting formalism you are using and to consider whether the nitrogen lone pair is participating in the bonding. These are summarized below. Notice the isoelectronic nature of the imido and the oxo ligand:
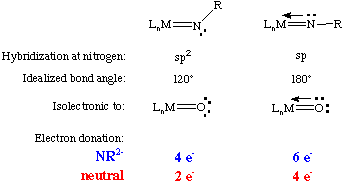
The M-N-R bond angle can, in most cases, be considered as an indicator of the degree of pi-donation from nitrogen. Imido ligands are such good pi-donors that the overwhelming majority of imido complexes are linear or near-linear (bond angles from 150 to 180 degrees) six-electron donors. The only circumstances under which an imido ligand is not linear are:
Imido ligands are of interest not only for their role as ancillary ("spectator") ligands, but for the potential group transfer of the imido unit to an organic moiety
Two excellent resources (with dozens of original literature references) are:
Purchase this text on-line. Also includes articles on slow proton-transfer reactions, oligopyridines in metallosupramolecular chemistry, ternary transition metal sulfides, and Pd-catalyzed oxidation reactions.